The European Health Data Space (EHDS) Regulation was published in the Official Journal of the European Union on 5 March 2025 and will enter into force on 26 March 2025. This new EU law establishes a common framework for the use and exchange of electronic health data across all Member States.
It is a cornerstone of the European Health Union initiative and the first EU-wide data space dedicated to health, aiming to enhance patient control over health records and enable the safe sharing and reuse of health data for healthcare and research purposes.
What the EHDS Regulation Entails
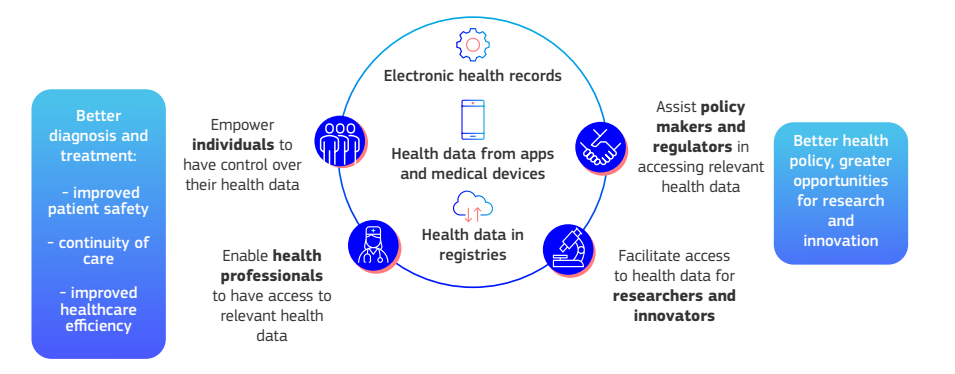
The EHDS Regulation introduces uniform rules and infrastructure to facilitate cross-border digital health services and data exchange within the EU. Its key objectives include strengthening individuals’ rights to access and control their personal electronic health data and establishing conditions for certain health data to be reused in the public interest, such as for research, innovation, public health policy, and regulatory activities.
In practice, this means EU citizens will be able to retrieve and share their health records (e.g. patient summaries, e-prescriptions, lab results) with healthcare providers across Member States, while having assurances of privacy and security. The regulation also creates a health-specific data governance framework: each Member State must appoint a digital health authority to oversee primary use (healthcare delivery) of data and a health data access body to manage secondary use (research and policy) requests. Source
.
The EHDS sets out technical standards for electronic health record (EHR) systems to ensure interoperability. All EHR systems in the EU will be required to comply with a European EHR exchange format so that health data can be understood and accepted uniformly across different countries and IT systems
This harmonized approach is intended to break down existing barriers created by varying levels of digitalization in national healthcare systems, and to support a single market for digital health products and services
The regulation also explicitly complements existing EU data laws, aligning with the General Data Protection Regulation (GDPR) and building on frameworks like the Data Governance Act and forthcoming Artificial Intelligence Act to maintain consistency across the regulatory landscape.
Under the EHDS, individuals will benefit from new rights and tools regarding their health information. They can expect fast, free access to their own electronic health data and the ability to easily share these records with doctors anywhere in the EU..
Patients will have more granular control – for example, they may add personal health notes, restrict access to certain parts of their records, see a log of who accessed their data, request corrections, and opt out of having their data used for secondary purposes like research. Strong security and privacy protections are mandated by default, given that health data is highly sensitive.
These measures empower patients while fostering trust in the digital health ecosystem.
Reactions from Key Stakeholders
Healthcare Providers: Hospitals and healthcare organizations support the EHDS but warn of complex implementation. The European Hospital and Healthcare Federation (HOPE) highlights the need to consider national differences in digital infrastructure.
Significant IT upgrades, interoperability efforts, and staff training may strain already tight budgets. Providers stress that real-world experience must guide implementation, with clear communication and stakeholder input.
HOPE emphasizes that the EHDS can’t be a top-down shift—frontline insights are crucial. The sector calls for realistic timelines and support to prevent widening digital inequalities in healthcare.
MedTech and Pharma Industry: A group of 39 European health leaders has welcomed the EHDS regulation’s publication in the EU’s Official Journal. They emphasize the need to quickly set up a well-funded, inclusive Stakeholder Forum to support smooth and effective implementation. They urge policymakers to:
- Establish the Stakeholder Forum early for active involvement
- Ensure fair funding and invest in capacity building
- Strengthen collaboration between the Stakeholder Forum and the EHDS Board
Similarly, pharmaceutical companies, through the European Federation of Pharmaceutical Industries and Associations (EFPIA), praised the regulation as a “significant step forward” in leveraging health data to advance research, innovation, and care delivery across Europe.
Significant IT upgrades, interoperability efforts, and staff training may strain already tight budgets. Providers stress that real-world experience must guide implementation, with clear communication and stakeholder input.
HOPE emphasizes that the EHDS can’t be a top-down shift—frontline insights are crucial. The sector calls for realistic timelines and support to prevent widening digital inequalities in healthcare.
Patient Advocacy Groups: European patient organizations have generally endorsed the EHDS Regulation for its potential to empower patients and improve continuity of care. The European Patients’ Forum (EPF), an umbrella group, points out that the primary goal of EHDS is to give patients seamless access to their health records and enable their medical information to travel with them across borders, which can lead to safer and more informed treatment
Regulators and Data Protection Authorities: European regulators have underscored the importance of privacy compliance and clarity as the EHDS moves forward. The European Data Protection Board (EDPB) and European Data Protection Supervisor (EDPS), in a joint opinion during the legislative process, welcomed the EHDS’s intent to strengthen individuals’ control over health data but also cautioned that the scheme must strictly uphold GDPR principles.
They drew attention to potential risks in the area of secondary data use, noting that while reuse of health data can yield public benefits, it “is not without risks for the rights and freedoms of individuals”. Source
Data protection officials have pressed for clear definitions and safeguards to avoid confusion between rights under EHDS and existing GDPR rights, warning against any legal uncertainty that might undermine data subjects’ protections
National health regulators and the European Commission have also highlighted practical challenges: for example, differences in national laws and health IT systems will need careful alignment. The Commission has stressed that additional implementing acts and technical specifications will be developed to guide how data sharing, consent, and security should work in detail, to ensure uniform enforcement of the rules across all Member States
EU Health and Food Safety has welcomed the EHDS highlighting its potential to drive digital innovation, enhance research, and empower citizens through e-Health. It states in the press release, ”The EHDS represents a major leap forward in strengthening the resilience of Europe’s health systems, ensuring they can tackle today’s most pressing challenges such as an aging population and workforce shortages.”
Implementation Challenges
While the EHDS Regulation is set to launch a new era of EU-wide health data exchange, stakeholders agree that realizing its full potential will require overcoming several implementation challenges.
One major challenge is technical and infrastructural readiness. HOPE and Data Saves Lives raise concerns over member states having varying digitized health systems and all of which do not have compatible electronic record systems in place. Building the required infrastructure (such as national health data hubs, secure data exchange platforms, and interoperable EHR systems) will demand significant investment, expertise, and political will. Health organizations warn that the upfront costs of this digital transformation – upgrading hospital IT systems, recruiting or training specialized personnel, and maintaining robust cybersecurity – could be substantial, and they call for adequate funding and support at both EU and national levels.
Another challenge lies in harmonizing legal and regulatory frameworks. Aligning with GDPR and national laws, ensuring interoperability and building trust are major challenges. Clear guidance on consent, IP protection, and data sharing is needed. Standardizing health data exchange is crucial to avoid fragmentation. Success depends on strong governance, funding, and clear communication to earn public and healthcare provider trust.
Transition Timeline and Key Milestones
The EHDS Regulation enters into force on 26 March 2025, marking the start of the transition phase.
By March 2027, the European Commission will adopt implementing acts detailing technical and procedural rules.
From March 2029, Member States must enable cross-border exchange of key health data (e.g., patient summaries, e-prescriptions) and allow secondary use of electronic health records.
By March 2031, expanded data types, including medical images and lab results, must be integrated.
Finally, by March 2034, international entities may gain access to EHDS for secondary use, completing the phased implementation.
Refer Frequently Asked Questions on the European Health Data Space to get more clarity on this.
Wrapping Up
The EHDS creates a unified framework for health data access and exchange across the EU, ensuring both privacy and innovation. Patients will have seamless digital access to their health records, while researchers and MedTech companies can use anonymized data to drive healthcare advancements. Strong data protection measures will safeguard privacy and security.
With the transition phase starting on 26 March 2025, healthcare providers and industry players must prepare for compliance as the EU Commission finalizes implementation guidelines. This marks a major step toward a more connected, data-driven healthcare system in Europe.
Learn how MedQAIR can help you navigate EHDS compliance and make the most of this new health data era.